
Aims & Outcomes
Primary Outcomes
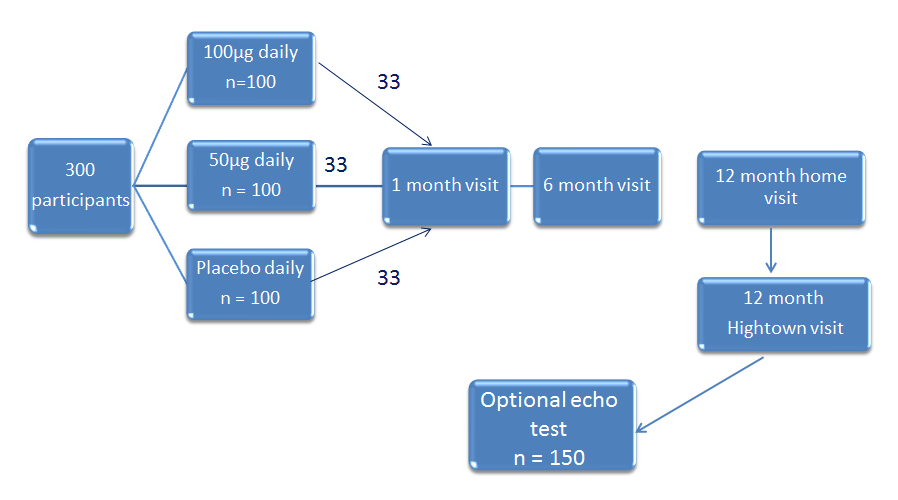
The aim of BEST-D is to establish the most appropriate dose of vitamin D to assess in a large outcome trial. The primary outcomes are:
- The difference in the proportion of individuals with 25(OH)D blood levels of >90 nmol/L between those alloctated 100 vs 50 µg vitamin D daily at the end of the study.
- The difference in mean 25(OH)D blood levels between those allocated 100 vs 50 µg vitamin D daily at the end of the study.
Secondary Outcomes
Analyses of the effects of daily vitamin D3 100µg vs placebo, vitamin D3 50µg vs placebo, and between the two doses of vitamin D on:
- Mean levels of 25(OH)D during follow-up
- Proportion of patients with normal PTH at 6 and 12 months
- Differences in the change from baseline in various inflammatory markers at 6 and 12 months (CRP, BNP and inflammatory cytokines)
- Differences in change from baseline in BP, heart rate and arterial stiffness at 6 and 12 months
- Differences in the change from baseline in cholesterol levels and cholesterol sub-fractions and apolipoproteins at 12 months
Exclusion Criteria
- Nursing home residents
- Regular use of vitamin D supplements (>400 IU daily)
- Use of alendronate, risedronate, zoledronic acid, parathyroid hormone, or calcitonin
- Medically diagnosed dementia, history of hypercalcaemia, hyperparathyroidism, lymphoma, sarcoidosis, active tuberculosis, or renal stones
- Poor compliance with clinic visits or with medication
- History of alcohol or substance misuse
- Medical history that may limit participation for the duration of the study (e.g. terminal illness)
- Prescribed calcium supplements.










