
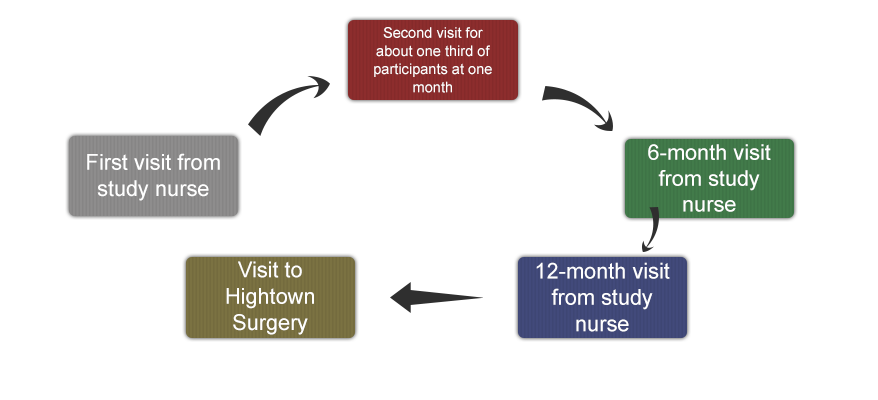
Study Timetable
Step 1
First visit from study nurse
Nurse checks your suitability, records clinical measurements and collects a blood sample. If you agree to take part, you will be asked to start taking two study capsules daily for one year.
Step 2
Second visit for about one third of participants at one month
About 100 participants will be asked to provide a blood sample after one month of study treatment.
Step 3
6-month visit from study nurse
Participants will be visited at home by a study nurse for clinical measurements, blood collection and to supply further capsules.
Step 4
12-month visit from study nurse
Participants will be visited at home by a study nurse for clinical measurements and blood collection.
Step 5
Visit to Hightown surgery
Participants will visit Hightown surgery for a bone density scan and to have additional measurements of physical function. A sample of participants who have opted to participate in the heart study and are invited, will have the echo test.
Total study duration: 12 months
Participants are selected randomly (by chance) to receive either 100mcg, or 50mcg vitamin D or matching dummy capsules.
The Vitamin D Capsules/Placebo


- Vitamin D helps the body to absorb calcium and phophorous and is important for keeping bones healthy. It may also have other health benefits.
- Low blood levels of vitamin D are common in older people, especially in the winter.
- It is not clear whether vitamin D supplements should be used routinely to increase vitamin D levels. The purpose of this study is to find out what daily dose of vitamin D is needed by older people to keep vitamin D levels at the peak summer levels seen in younger adults.
- One third of those taking part will be given 100 mcg vitamin D daily; one third will be given 50 mcg vitamin D daily, and one third will have dummy capsules which look the same. Neither you, nor the study nurse or your doctors will know which treatment you are on; this will be decided randomly (like tossing a coin).
- You will be asked to take two capsules of study treatment daily for one year and see the study nurse on at least three occasions over this period.
- The results of BEST-D will help with planning of a large study to test whether long-term vitamin D treatment protects against osteoporosis and other chronic health conditions.
- The capsules are very small and easy to take.
Devices

JAMAR handgrip dynamometer
The device is used to assess handgrip strength which reflects muscle strength. It is simple to use, accurate and gives reliable repeat measurements. In BEST-D, we will evaluate the effects of vitamin D supplements on handgrip strength.

Pulsetrace PCA2
This device uses a infrared sensor at the finger tip to assess the blood flow characteristics at your fingertips. The device is easy to use in large studies and provides an estimate of the stiffness of arteries. The effects of vitamin D on arterial stiffness after 1 year will be examined with the help of these readings (along with the Arteriograph below) in BEST-D.
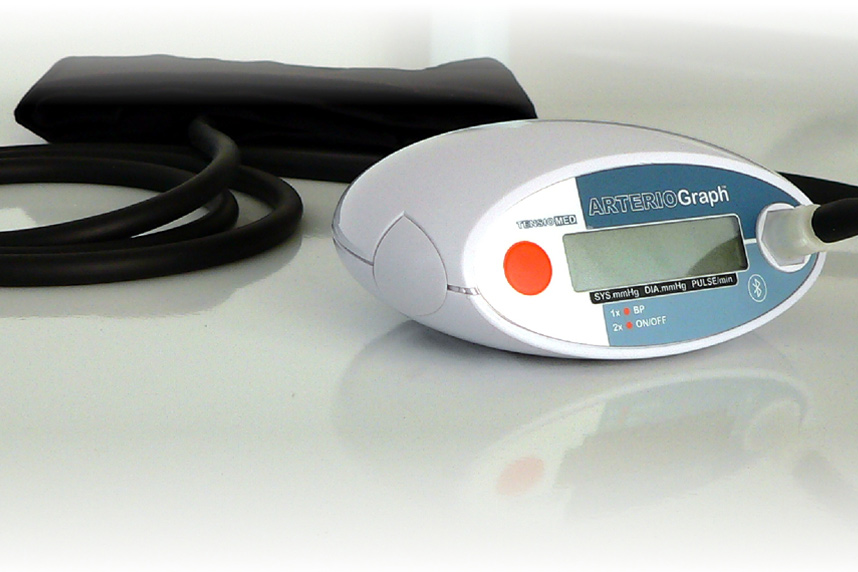
Arteriograph
The Arteriograph measures blood pressure, your pulse rate and records the pulse characteristics used to estimate arterial stiffness at your upper arm.
Frequently Asked Questions
Why have I been invited?
You are being invited to participate in the study because you are over the age of 65 years, and live in or around Banbury where the study is taking place. Your General Practitioner has agreed to your invitation.
Do I have to take part?
No, taking part is entirely voluntary. The study nurse will go through all information with you to confirm that you understand what the study involves. If you do decide to take part, you will be asked to sign a concent form. If you do do take part, you are free to withdraw from the study at any time without giving a reason and this will not affect your medical care in any way.
What will happen to me if I take part?
If you decide to join the study, you will be asked to take two small capsules daily for one year. Each capsule contains either 50 micrograms (mcg) of vitamin D or plain corn oil which is used as a dummy or placebo capsule. We hope to include about 300 older people in this study with 100 taking each dose (100mcg or 50mcg vitamin D or placebo).
If you choose to join the study, the dose that you would take will be decided randomly, like tossing a coin. This helps to make sure that the groups are as similar as possible except for the dose of vitamin D being taken. Neither you, nor the research nurse or doctor will know which treatment you are taking. This information will be stored securely on a computer at Oxford University and can be made available to your doctors if medically necessary. This is known as a double-blind randomized trial.
The study involves a study nurse visiting you at home on three or four occasions over one year and a single visit to Hightown Surgery at the end of the year. An appointment for a study nurse to visit will be made by telephone. The first visit will take about one and a half hours, but the 6 and 12 month visits should last less than one hour.
What happens at the study visits?
At the first visit, the nurse will ask about your medical history, diet and use of any prescription or over-the-counter drug treatment. This information will be entered directly onto a computer.
- If you are suitable for the study and interested in taking part, the nurse will explain what is involved and give you a chance to ask any questions. If you are willing you will be asked to sign a consent form and given a copy to keep.
- The nurse will measure your height, weight and hand-grip strength, blood pressure and a measure called "arterial stiffness". Arterial stiffness is measured as part of taking your blood pressure and also by using a finger probe (a small clip-on device); both are simple and painless.
- The nurse will take a 30ml blood sample (about 2-3 tablespoonfuls) to measure blood levels of vitamin D and other markers relevant to health and disease. One of the blood samples will be used to test genetic markers in future analyses. You are free to opt out of participation in any genetic analyses and you can indicate this on the consent form.
- The nurse will then give you two bottles of study treatent and ask you to take one capsule from each bottle every day. The capsules are small and contain either 50mcg of vitamin D, or plain corn oil without vitamin D (i.e. dummy capsules). You will be given enough capsules to last about 6 months.
At the end of the visit, you will be given an appointment for the next visit. The information collected by the study nurse on the computer will be transferred to the Clincial Trial Service Unit at Oxford University where it will be stored securely in a database.
Some participants will be asked to have an extra blood sample (about two tablespoonfuls) collected at one month after the first visit. This visit should only take around 15 minutes.
What will I have to do?
Your main responsibility will be to take the capsules regularly and let the research team know if you have any problems related to the study. All your other treatments and daily activities can continue as usual.
What substance is being tested?
The substance being tested is vitamin D, in form of cholecalciferol (also known as vitamin D3), taken by mouth. The dose of vitamin D used in this study (either 100mcg or 50mcg daily) is higher than the doses used in previous studies. However, some studies have safely used a higher dose of 2500 mcg (all in one dose) of vitamin D given every few months for several years. Millions of people around the world take vitamin D supplements which can be easily bought over-the-counter and are considered safe, but the typical doses are only 10 to 20 mcg daily.
What are the side effects and risks of taking part?
Very rarely, a high intake of vitamin D leads to high blood levels of calcium, which can cause people to feel unweel. Typically, they become thirsty or pass more urine than usual, feel sick or vomit, or have dizziness or headaches or become confused. However, any symptoms should disappear as soon as the vitamin D is stopped. Nevertheless, the doses of vitamin D used in this study are thought to be safe and not expected to raise blood calcium levels above the normal range. In the unlikely event of your becoming unwell, your calcium level will be checked. If at any time you think you are having side effects from the study medication, you can contact Dr Hin, or one of the study nurses at Hightown Surgery (01295 270722) or one of the CTSU doctors for advice (24-hour FREEFONE 0800 585323).










